How to Measure Salinity
Salinity is the measurement of the total dissolved materials in a solution. It is a mass per unit volume measurement. For example, seawater has a mass of around 35 grams of salts dissolved in a volume of one liter.
Boil off all of the water
Early oceanographers and limnologists (freshwater scientists) had to measure the amounts of salt in water by evaporating all of the water away from the samples. This was time intensive and prone to errs. Over time engineers and scientist came up with more ways to determine salinity.
Measure the density
If you have ever brewed beer you may have used a hydrometer measure how much sugar is in the wort. A hydrometer is a closed vessel that has a weighted bottom. The long end of the vessel is calibrated with gradations that are visible through the glass. A liquid that is more dense (seawater) will push the hydrometer further UP and out of the water, where a liquid that is less dense (freshwater) the hydrometer will sink further DOWN into the water.
Measure electrical conductivity
The most accurate way we measure salinity now is through a process called “conductivity”. This is a type of proxy measurement, where we measure one thing, to get a good estimate of another.
How do conductivity meters estimates salinity?
A conductivity meter measures the amount of electrical charge an aqueous solution can carry, or conduct. Pure water is a poor conductor of electricity, but when ions are dissolved in the water the ability to pass a current increases increases. . The conductivity meter uses a probe to measure conductivity of a solution. ... Depending on the concentration of ions in the solution, the conductance is either high or low, which results in a fast or slow current reading. The relationship between ions in water and conductivity is predictable and so we can use precise instruments estimate ‘salinity’ by measuring conductivity.
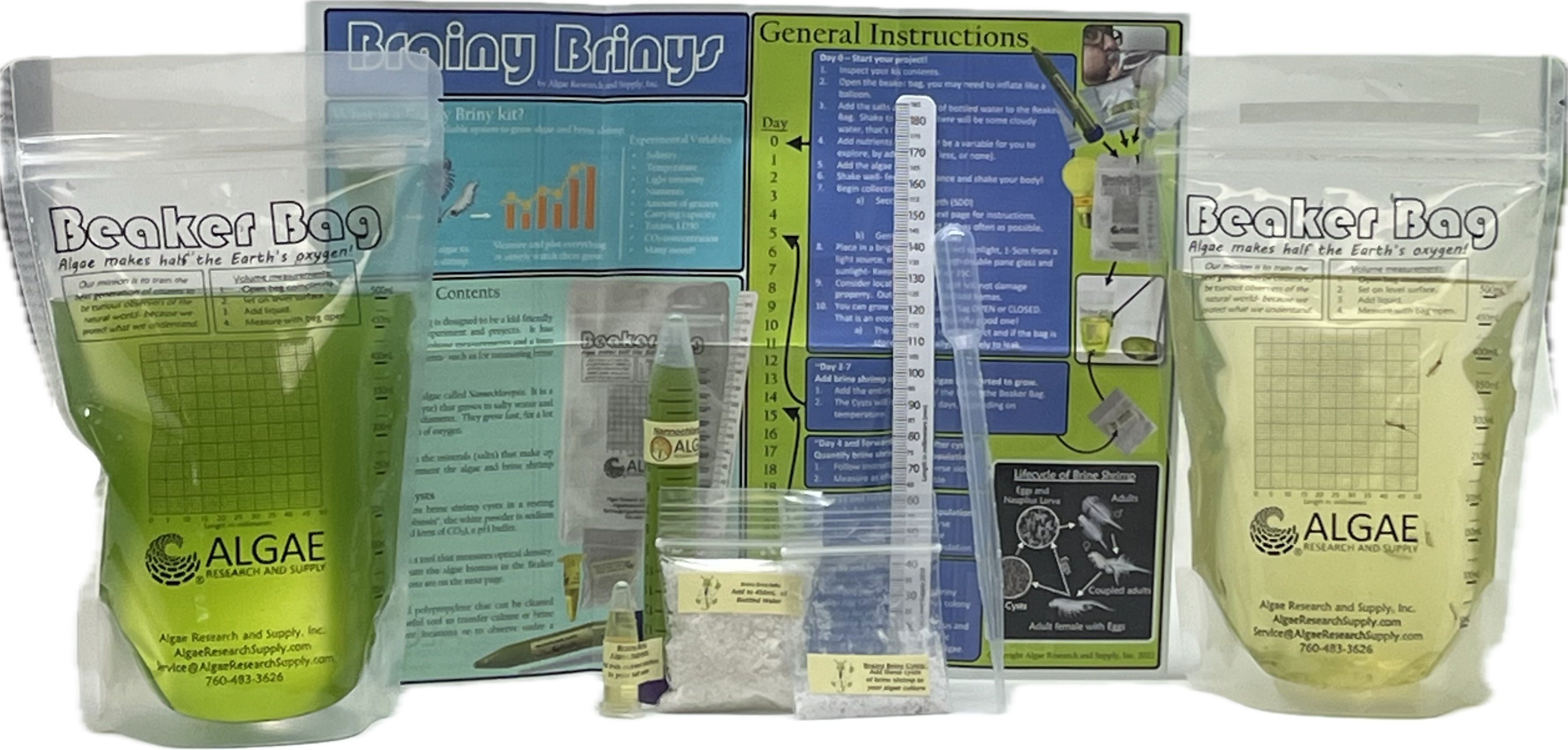
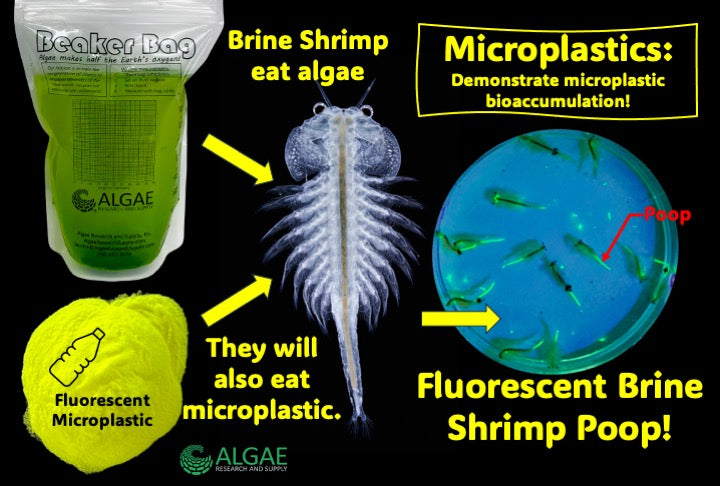
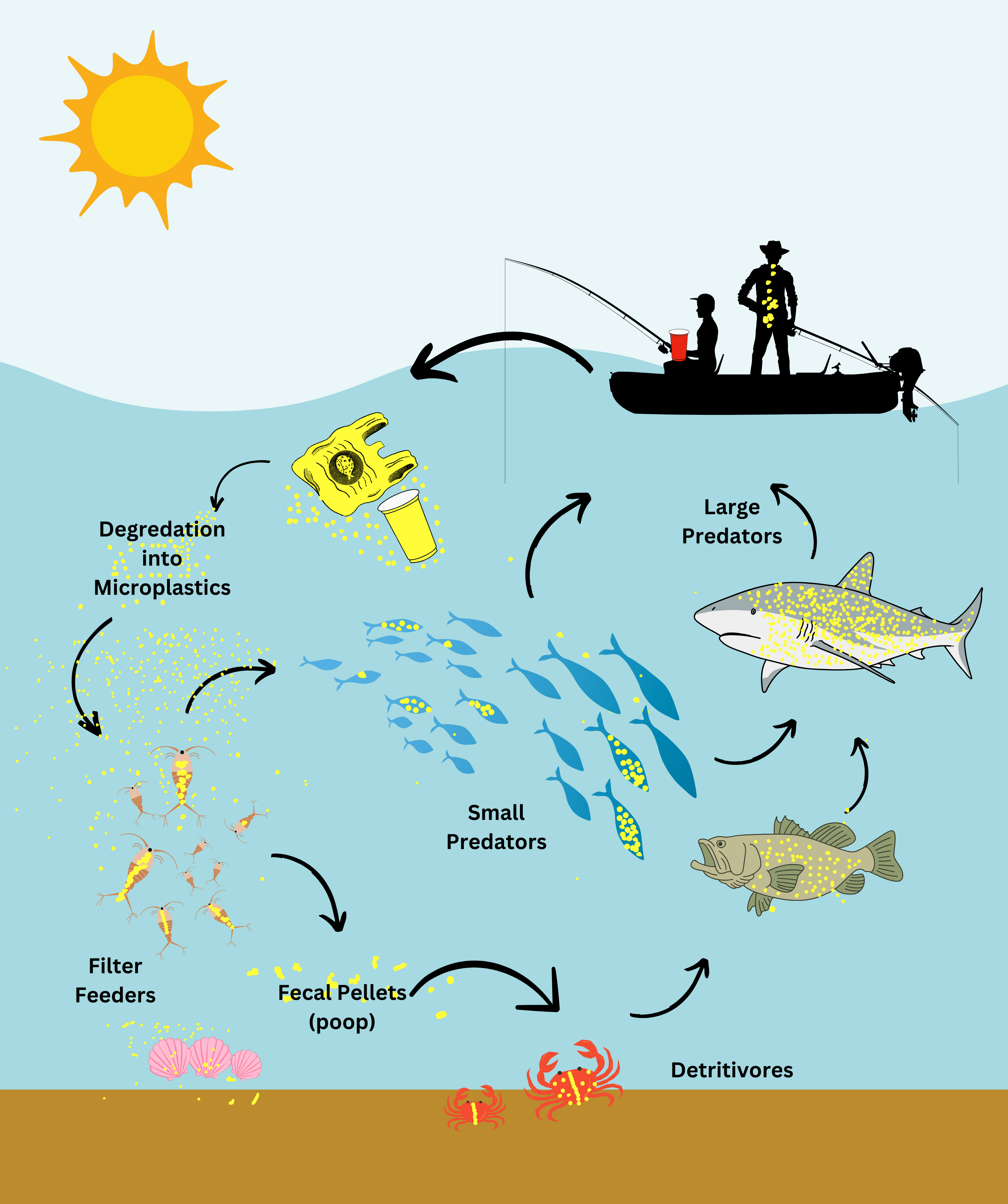
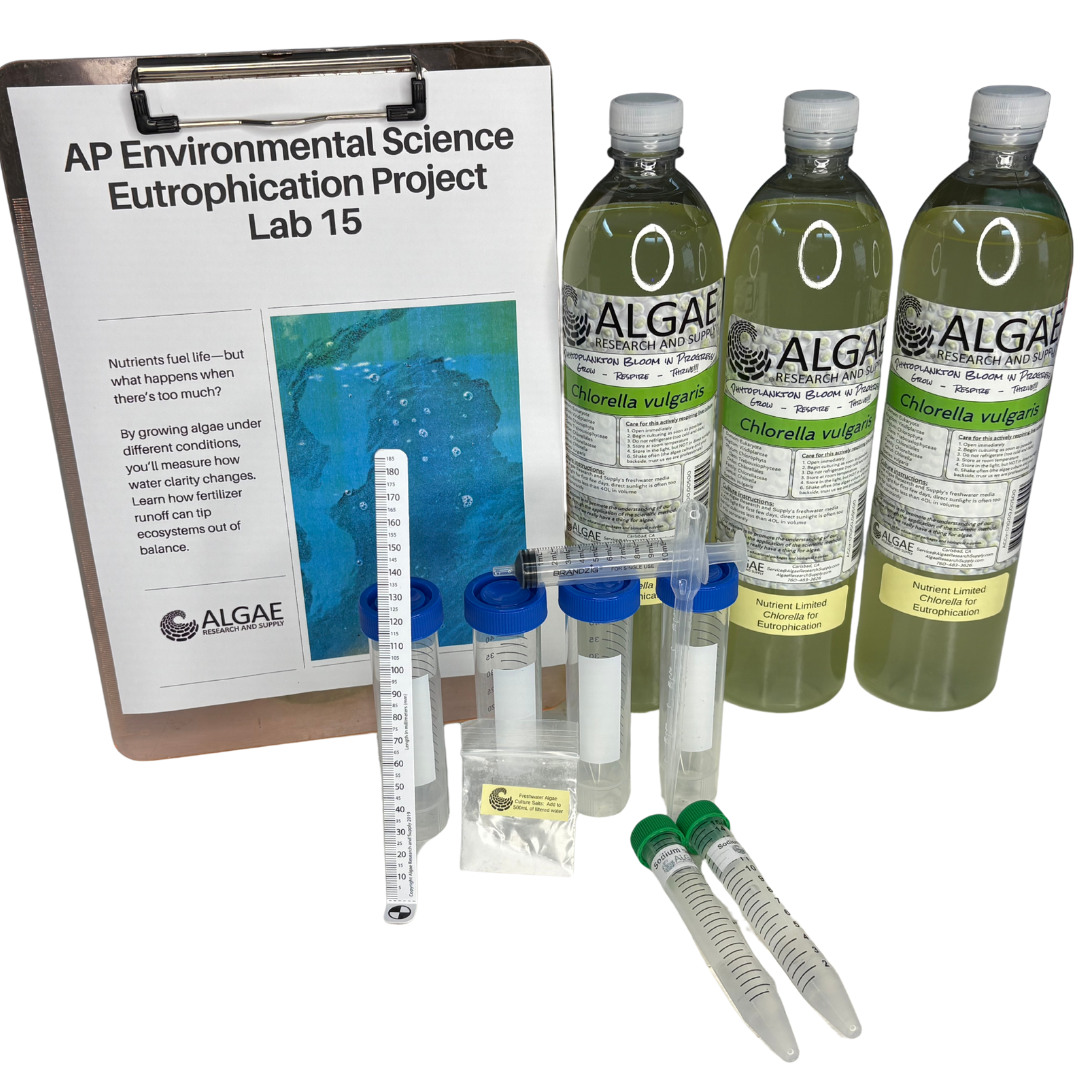
Ignite curiosity and foster hands-on learning with our comprehensive algae experiment kits. Designed for educators, students, and science enthusiasts, each kit provides all necessary materials to explore the fascinating world of algae. From culturing diverse algal species to conducting experiments on growth conditions, these kits offer a practical approach to understanding ecological and biological concepts. Ideal for classrooms, science fairs, or personal exploration, our kits make the study of algae accessible and engaging for all ages.