Measuring Growth
|
Measuring Growth |
Algae grows fast!!! Most strains of microalgae grow faster than any terrestrial plant. But what does it mean to grow. A growth rate is a change in biomass per change in time. Below is an examination of biomass measurements and how to express growth rates.
|
Estimating Biomass |
Estimating the amount of biomass in a system is often an important first step in understanding how matter and energy move between populations.
Weighing Biomass:
This is sometimes the best way to determine biomass, simply weigh it! *Weighing involves filtering, which is not a good thing to do if you want to keep the algae alive. So it is called a “destructive method” for quantification. For non-destructive methods look at the Secchi stick.
Wet Weight: (our favorite weighing method) A best practice is to simply filter a volume of culture onto a pre-weighed filter pad. Subtract the mass of the pad from the algae and the pad and that is your wet weight. This is expressed as grams of wet weight per liter of media, (g-WW / L).
Wet weight = (Mass of pad) - (Mass of pad and filtered algae)
Dry weight: (our favorite weighing method) More accurate than wet weight biomass measurements, dry weight is what most algae growers will talk about when describing the amount of biomass in their ponds. It is identical to the wet weight process, except the pad is often washed with fresh water to remove salts then oven dried at 100C until all the water is driven from inside of the cells (usually a half hour). The final mass is then determined immediately after weighing so moisture from the air does not reabsorb into the algae. (g-DW / L)
Dry weight = (Mass of pad) - (mass of oven dried algae and pad)
Ash-Free Dry Weight: Some researchers get drunk with informational-power and want to know exactly how much organic matter is in the biomass- the ash free dry weight. This process helps you determine how much “alive stuff” is in your algae, or conversely, how much non-biological cellular material the cell has in them. A good example of when to use this method, is to estimate the mass of silica in a radiolarian cell wall or a diatom frustule, or calcium carbonate in a haptophyte. Can you imagine an experiment where you varied the amount of silica available to a diatom culture in an effort to learn if their frustules got thicker with extra silica? To perform this process, you perform the dry weight procedure, then burn off all the biomass in a furnace and weight again. (g-AFDW / L)
AFDW = (Mass-post furnace) - (Dry Weight)
How dense in dry weight are natural systems?
- Open ocean: ~0.0001g/L
- Coastal oceans: ~0.01 g/L
- Coastal and lake algae blooms: 0.1g/L
- Lab cultures in photobioreactors: 1 g/L
Counting Cells, hemocytometer:
Using a microscope and a specialized slide called a hemocytometer, you can count the number of cells on the slide. When a cover-slip is placed on the slide, the volume of liquid is fixed, and you can find the number of cells per area. By multiplying to the volume of the sample, you can estimate the population of cells.
The downside: You need a microscope with reasonably good optics and a hemocytometer ($30-300). Also you have to ‘math’ a little to make it work.
By Optical Density:
Using the Beer Lambert laws of optical density there are several ways that you can estimate biomass, the most easily used here are: turbidity-meter, spectrophotometer, and secchi stick.
The turbidity-meter and the spectrophotometer. They are meters that generate light, pass the light through a sample, then detect the light that has made it through the sample to a light meter. The difference is that a spectrophotometer can be adjusted to emit light across a range of the electromagnetic spectra and the turbidimeter uses a few wavelengths to estimate the turbidity of the water.
Downside: Spectrophotometry costs $3,500-50,000 per unit and is generally not portable for field use. The turbidiometer is often portable but still has a price tag in the hundreds of dollars
The Secchi Stick is our favorite meter, it is easy to use, nondestructive and can fit in your pocket with a cost of $2. The Secch Stick is a derivation of a very old tool used to determine turbidity in water. The target on the stick is lowered into the water until the user can not discern the pattern in the target. The depth in to the water when the pattern is not identifiable is called the Secch Stick Depth. A common assumption is that 3.1x the SDD is the 1% light level which is near the intensity of light where photosynthesis and respiration are equal (aka the compensation intensity).
This stick is great for use with students and is as accurate as a student-grade spectrophotometer.
Downside: Great, like really great for student work or cell cultivation, but for scientific publication dry weight or cell count are also required..
|
Growth Rates |
A growth rate is a change in biomass per change in time. There are different ways we can talk about growth, some that are more simple than others. Personally, I like the simple, but there is merrit to explaining the more complex.
Percent new biomass per day:
This is one of the simpler metrics. Simply compare the biomass on successive time points, usually days.
( biomass-day-2 - biomass-day-1) / (biomass-day-1) x 100
Example 0.010g/L on monday, 0.015g/L on tuesday (0.015-0.010) / (0.010) x100 = % growth
Specific Growth Rate (μ)
The specific growth rate (μ) of the microalgae is calculated using the equation μ = ln(N2/N1)/(t2 − t1), where μ is the specific growth rate, and N1 and N2 are the biomass at time 1 (t1) and time 2 (t2), respectively.
This is a unitless measurement.
Areal Growth Rate:
Areal growth rate is the new biomass per area per time. This is usually used to express growth in a pond or in the ocean. (mass per area time, g/m2s)
Doubling time:
The doubling time is time it takes for a population to double in size/value.
Next step ---> Harvesting
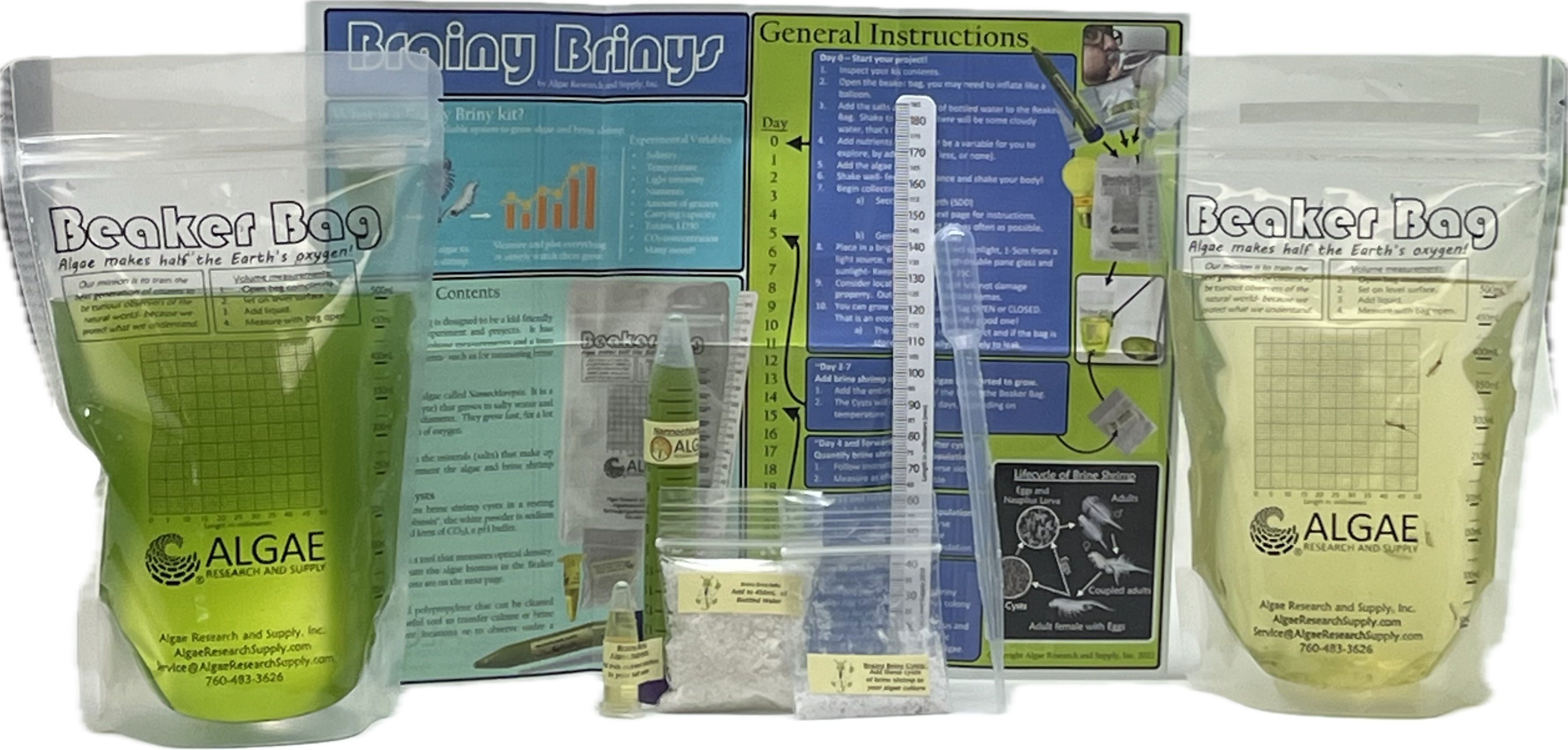
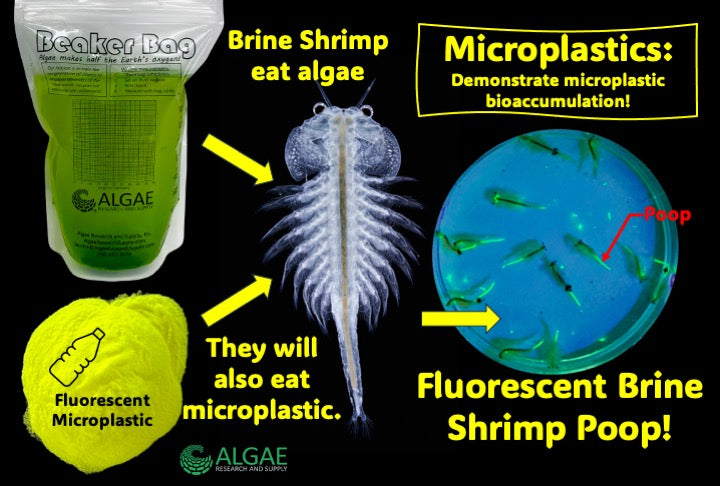
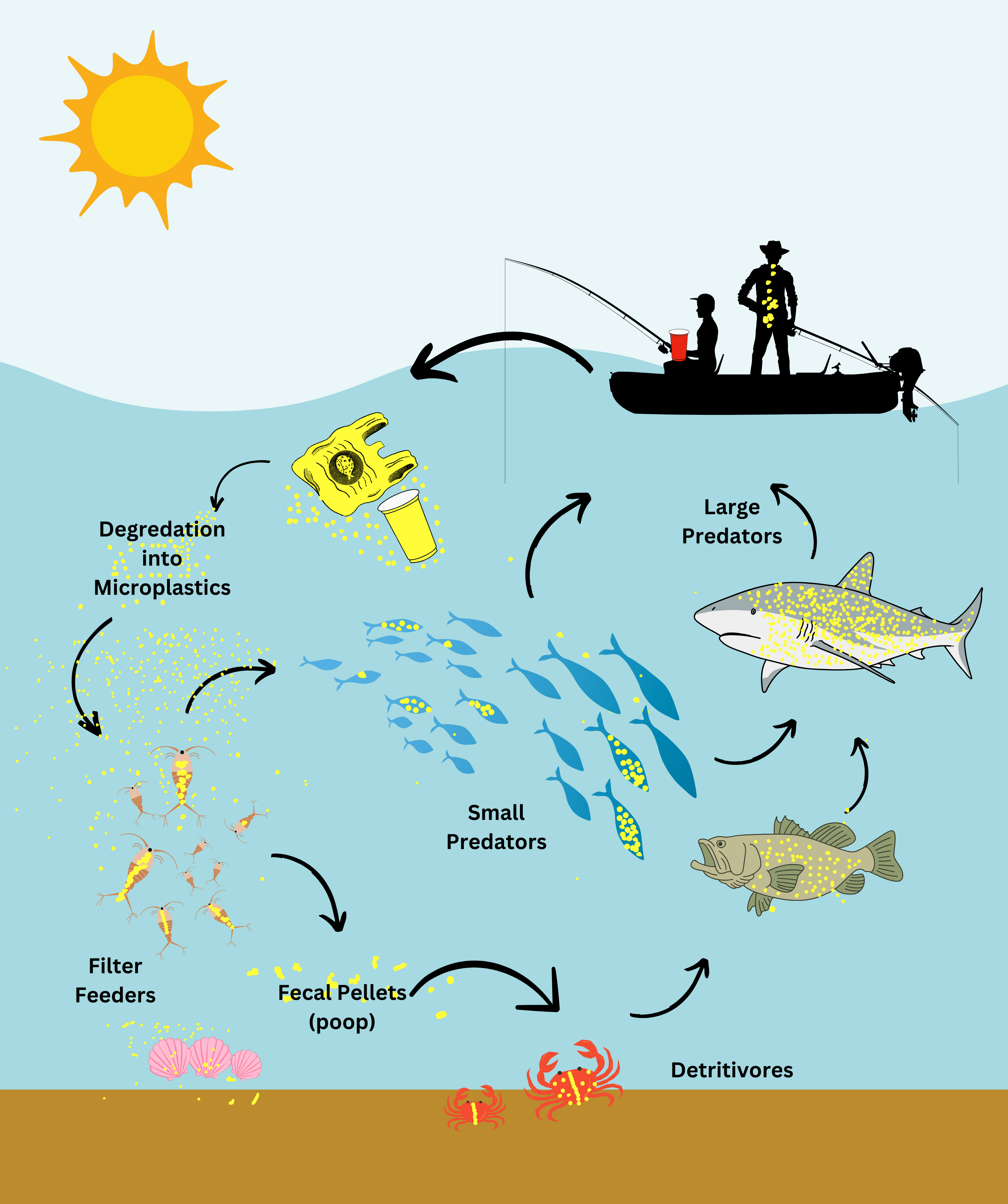
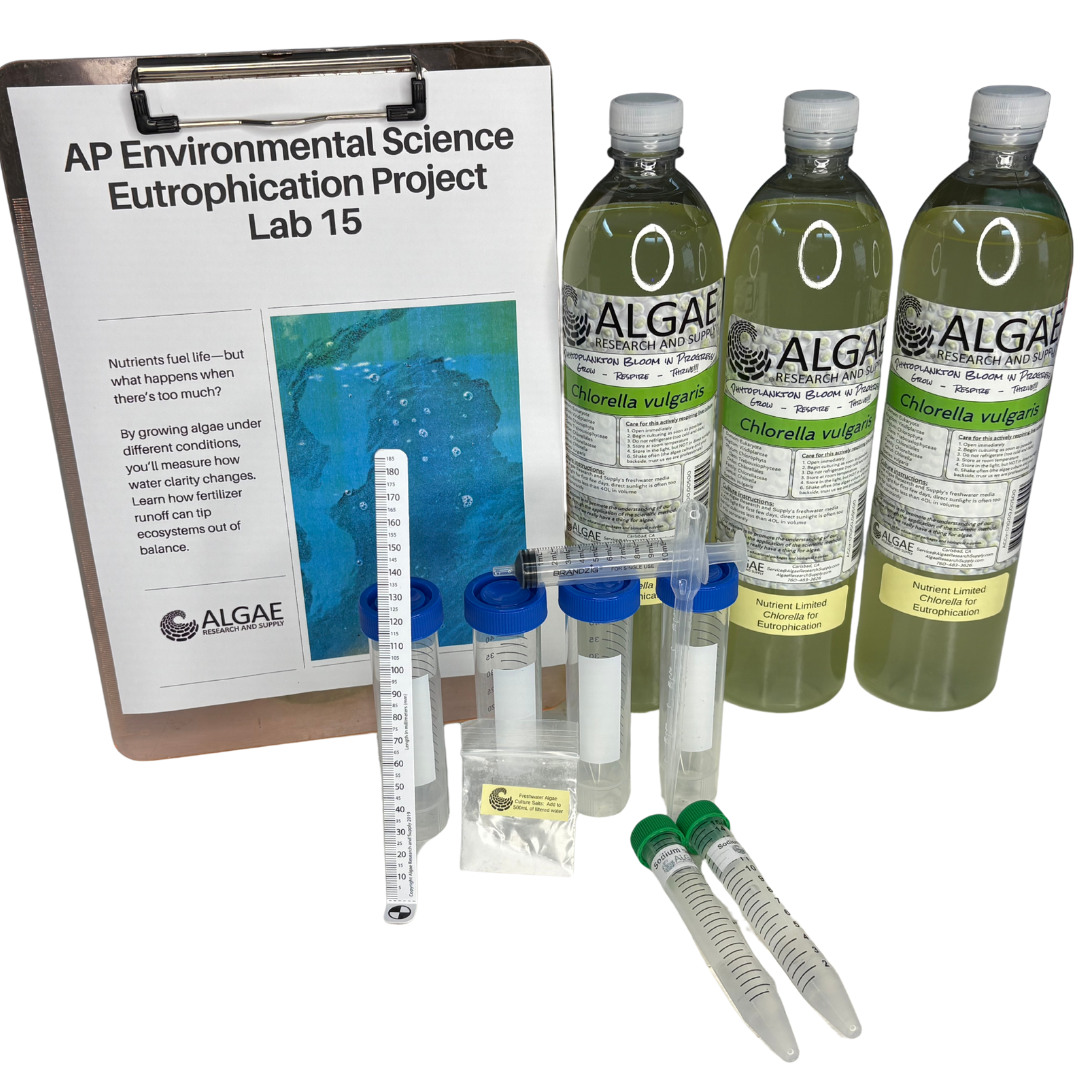
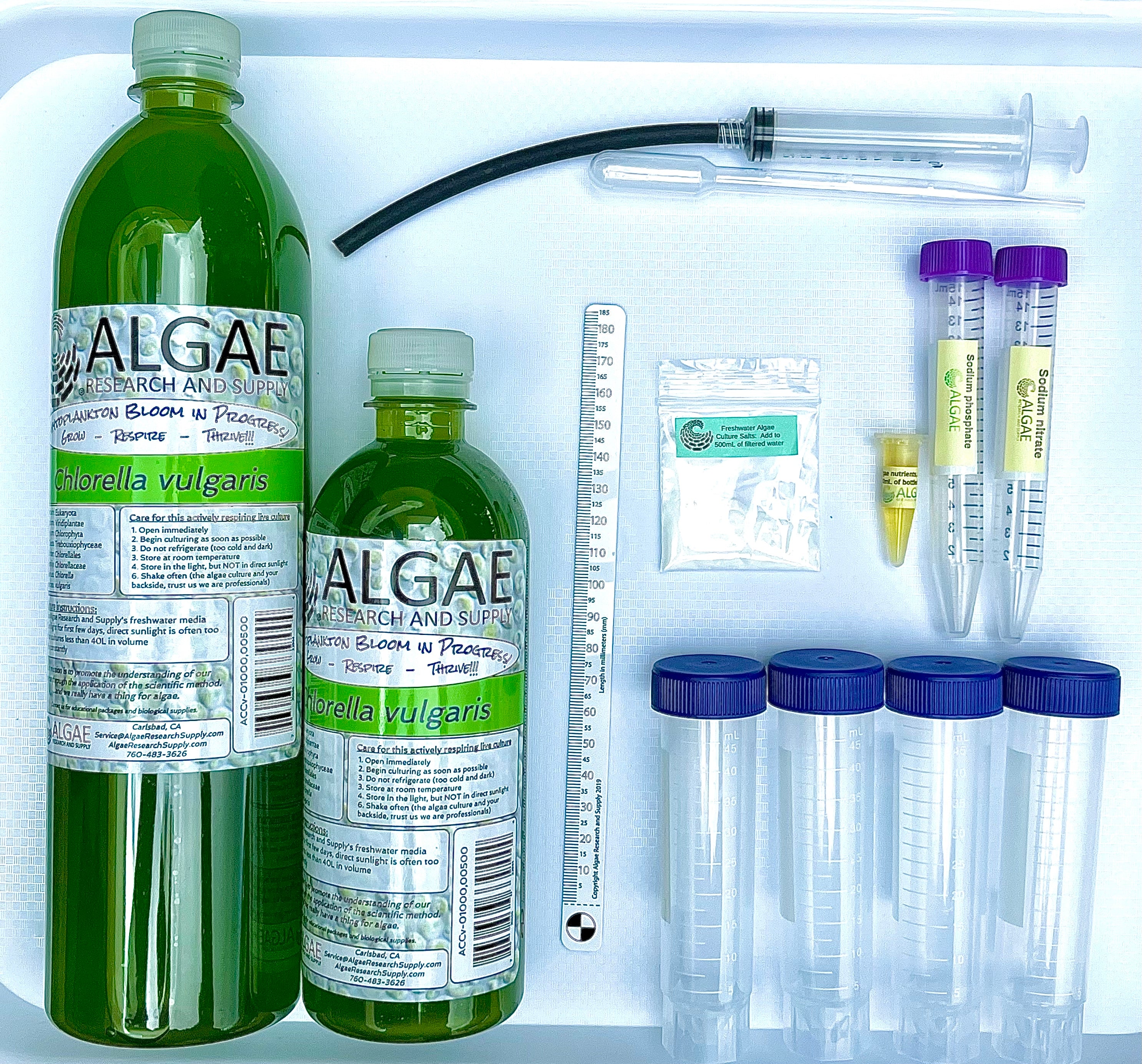
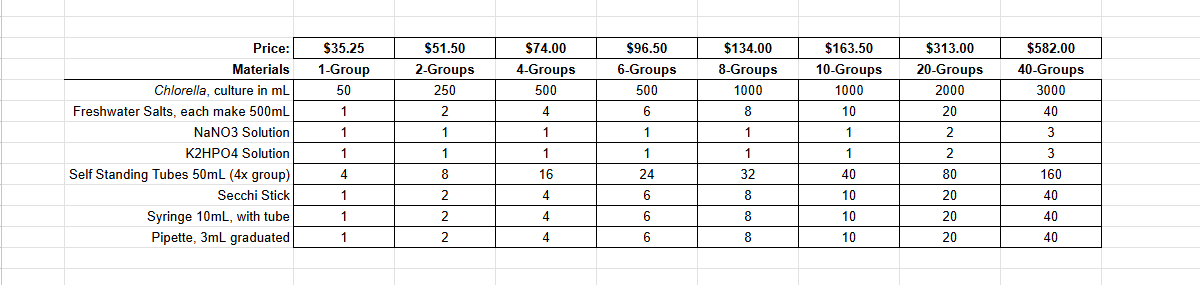
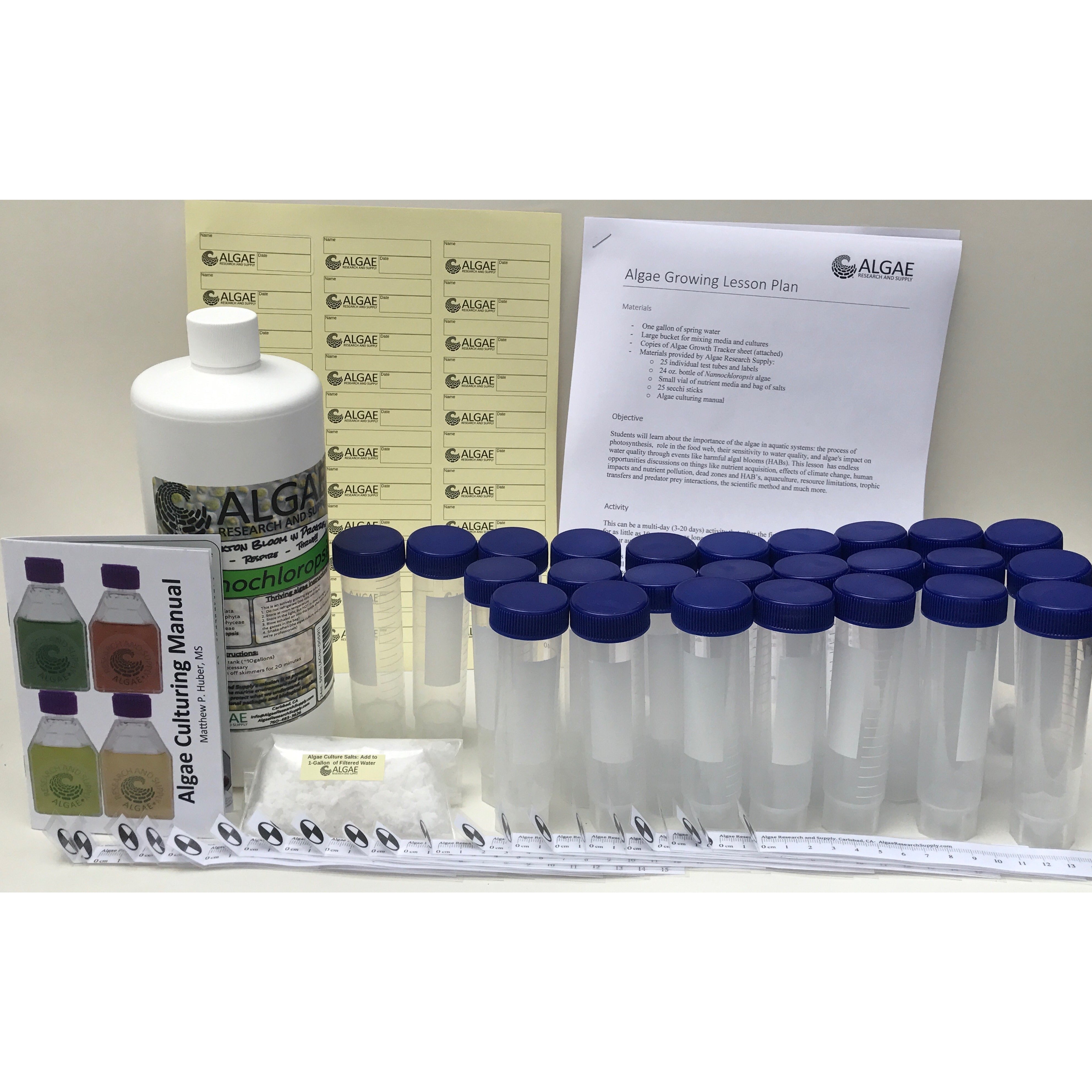
Ignite curiosity and foster hands-on learning with our comprehensive algae experiment kits. Designed for educators, students, and science enthusiasts, each kit provides all necessary materials to explore the fascinating world of algae. From culturing diverse algal species to conducting experiments on growth conditions, these kits offer a practical approach to understanding ecological and biological concepts. Ideal for classrooms, science fairs, or personal exploration, our kits make the study of algae accessible and engaging for all ages.