Algae Beads: Projects
Algae Bead Module: Lessons, Projects, and Experiments
Projects: Algae Beads
General NGSS Projects for Photosynthesis, Respiration, and Algal Physiology
Aligned for NGSS Standards:
HS-LS1-5 |
Use a model to illustrate how photosynthesis transforms light energy into stored chemical energy. |
HS-LS1-6 |
Construct and revise an explanation based on evidence for how carbon, hydrogen, and oxygen from sugar molecules may combine with other elements to form amino acids and/or other large carbon-based molecules. |
HS-LS1-7 |
Use a model to illustrate that cellular respiration is a chemical process whereby the bonds of food molecules and oxygen molecules are broken and the bonds in new compounds are formed resulting in a net transfer of energy. |
HS-LS2-5 |
Develop a model to illustrate the role of photosynthesis and cellular respiration in the cycling of carbon among the biosphere, atmosphere, hydrosphere, and geosphere. |
HS-PS1-5 |
Apply scientific principles and evidence to provide an explanation about the effects of changing the temperature or concentration of the reacting particles on the rate at which a reaction occurs. |
HS-PS1-6 |
Refine the design of a chemical system by specifying a change in conditions that would produce increased amounts of products at equilibrium. |
HS-LS4-4 |
Construct an explanation based on evidence for how natural selection leads to adaptation of populations. |
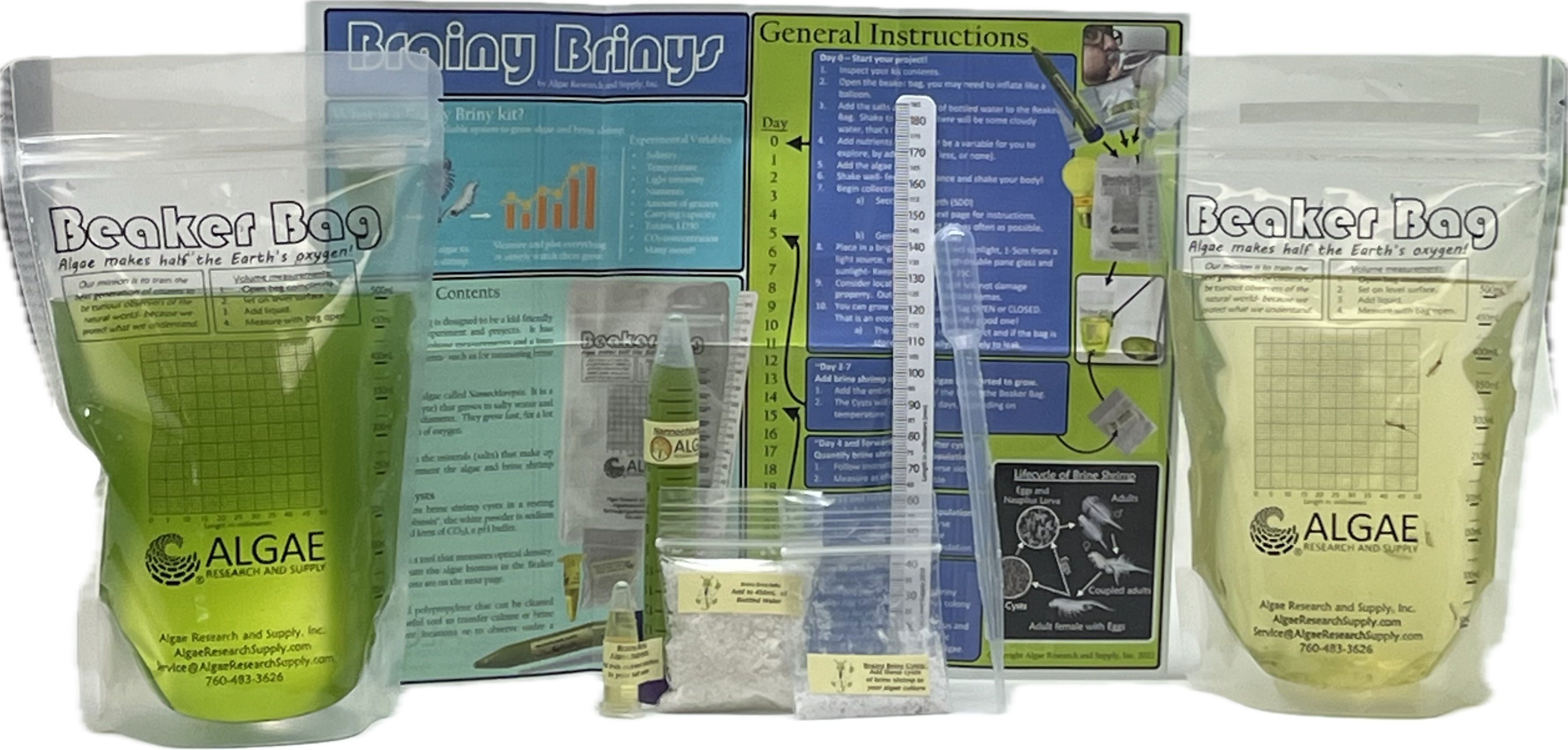
Make Your Own Algae Beads (A complete kit for growing algae, making algae beads, making pH indicator, and performing experiments.) (ARS sku: ABGK-BB-00050)
Copyright 2020 by Algae Research and Supply, Inc. Carlsbad CA. All rights reserved.
Algae Research and Supply, Inc.
Carlsbad, CA 92008
AlgaeResearchSupply.com
Table of Contents
Culturing Algae for Algae Beads
(Algae Bead Project 1 of 3) 4
Creation of Algae Beads
(Algae Bead Project 2 of 3) 8
Creation of Algae Bead Vials: pH Indicator Adjustments and Filling Vials
(Algae Bead Project 3 of 3) 11
How to use this Module:
This module contains standalone lessons, long-term projects, and experiments using microalgae as a model organism to teach aspects of respiration, photosynthesis, and cellular physiology. Much of the hands-on experimentation employs sodium alginate beads (ASR), similar to Petri agar, and provides a discrete investigation unit for students to manipulate. ARS provides pre-made beads and liquid cultures and instructions for making your own beads if you wish to do so!
Lessons are suggested materials and activities to educate on photosynthesis and respiration using algae beads. They are formatted into tables for easy processing and digestion. Each sub-lesson within a table contains suggested duration, content including links to videos, “Learning outcomes”, “let me prove it”. Each activity directs students to read the content and a learning outcome of that activity. After the students have read the paragraph of material, they are directed to answer the “let me prove it” questions that solidify the material they learned. Feel free to change and augment the lessons. Use them as a suggestion for presenting the relevant concepts to your class; add, expand, or remove topics according to your desired curriculum.
Projects below are long term and guided lab sessions that do not require hypothetical formulation. They teach students algal culturing and maintenance, as well as create stocks for future experiments. Project Worksheets are designed to act as an easy to use lab notebook, an essential component of any scientific endeavor.
Experiments are what they sound like, discrete lab sessions containing background information, lists of necessary material and methods, and a worksheet section for hypothesis-building and data collection. Teachers are encouraged to augment or add to these experiments to fit your desired curriculum.
|
Lessons
|
Projects |
Experiments |
Culturing Algae for Algae Beads
(Algae Bead Project 1 of 3)
|
Project Summary:
This project is to grow microalgae and concentrate it for making algae beads. If the culture grows well, it will have an estimated 0.2 to 0.5g dry-weight per liter of algae biomass in 15mL of media.
Time Estimates: Initial setup-10 minutes, followed by 1 minute per day for 14 days.
Learning Outcomes:
Classroom Prep: Divide class into lab groups if desired. See the materials list for the required components
|
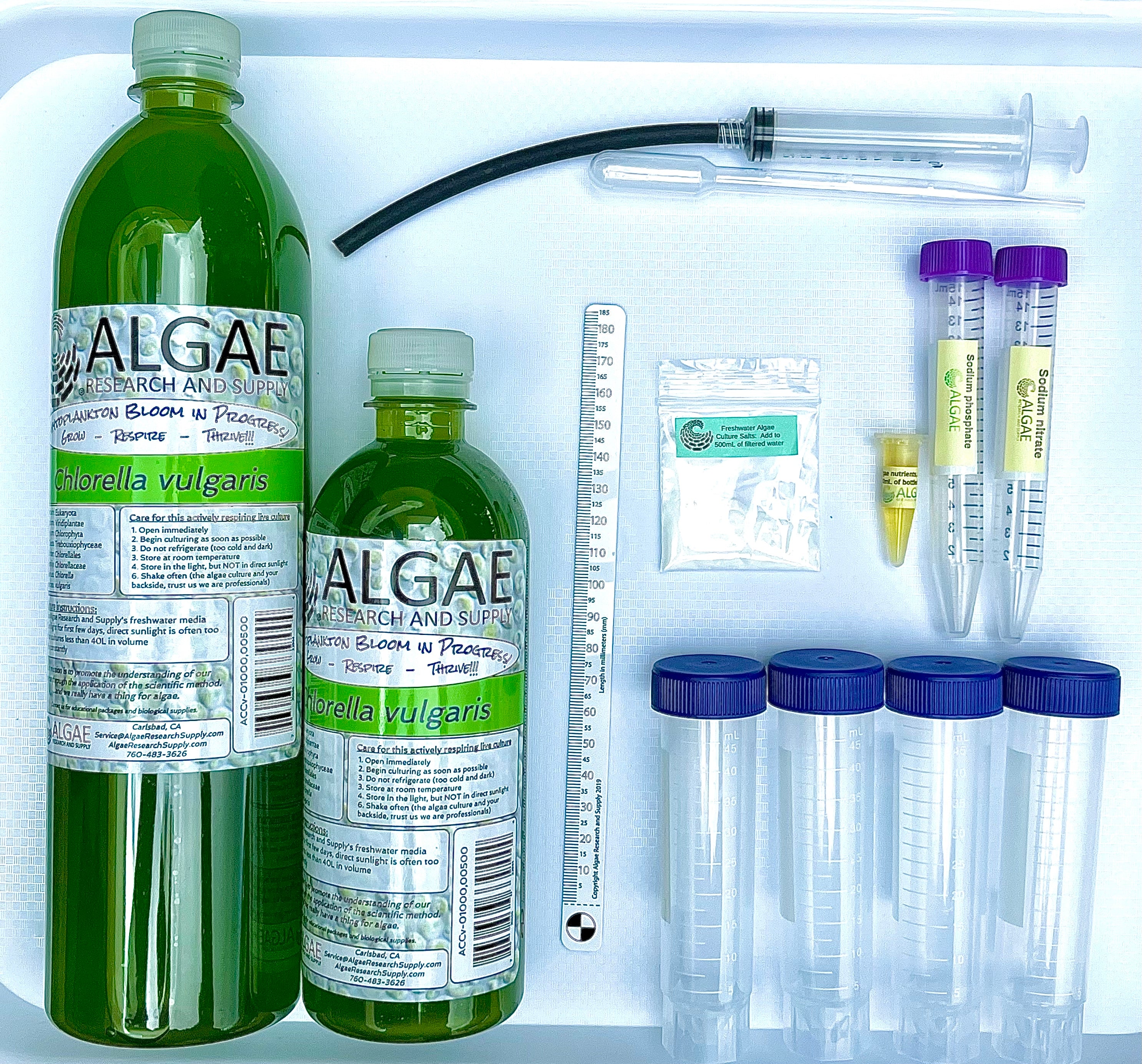
Materials
- Make Your Own Algae Beads (A complete kit for growing algae, making algae beads, making pH indicator, and performing experiments.) (ARS sku: ABGK-BB-00050)
- Beaker Bag, 500mL
- Algae Culture, 50mL
- Nutrients, 1mL
- Salts, Freshwater for 500mL
- Secchi Stick
- Bottle of water, 500mL (preferably bottled spring water, avoid tap water *chlorine*)
- Light source, CFL or LED
Procedure
- Make the media (this is the non-biological matrix that the algae live in… basically fertilizer spiked water!)
- Place in front of you
- Bottle of drinking water, 500mL
- Freshwater salt baggie
- Vial of nutrients.
- From your bottle of water, pour out ~50ml. This is to make room for the other components.
- Add the baggie of freshwater salts to the bottle of water.
- Add the 1.5mL vial of nutrients to the bottle of water.
- Shake the bottle to dissolve, shake your backside also. Trust us, we’re professionals.
- Inoculation with the algae starter culture
- Place in front of you
- The Beaker Bag. Open it by placing it to your face and exhaling, it will blow up like a balloon. Make sure it can stand up on its own.
- The bottle of media from the previous step
- The algae culture, 50mL Chlorella vulgaris
- Add 100mL of the media (from bottle of water in step one) into the Beaker Bag.
- Add the algae culture included in the kit to the beaker bag.
- In case of apocalypse, you can set aside a few mL of culture to make up for any unknown algae death. Add a splash of the nutrient media to it and put it in an out of the way place out of direct sunlight.
- Mix the bottle gently so that the culture homogenizes (until everything in the bottle is evenly mixed).
- You will add the rest of the media on day-3.
- Why are you waiting? The algae just arrived to you from our labs in Carlsbad, California. The algae need to acclimate themselves to your water, temperature, and light. This is called the “lag phase” in the algae batch culture lifecycle.
- Find a home for your algae.
- Light
- Place the bottle near your culturing light source;
- LED: Place the culture 1cm away, for a 6-20W led.
- CFL: Place the culture >5cm away. This placement is because CFL’s output more heat than LEDs and will “cook” your culture if not positioned appropriately.
- Do not place the algae in direct sunlight, as it provides too much heat and light to such a small volume of culture.
- If you absolutely must do this, be sure to shade ~80% of the incoming sunlight. Depending on your window, you may be able to reflect infrared energy (heat), but its best to err on non-algae murder.
- Confirm appropriate shading using your Google Science Journal light meter.
- Temperature
- Chlorella is tolerant of colder temperatures but generally does best between 22 and 27 degrees Celsius. Use a standard thermometer to confirm you are culturing your algae in an appropriate temperature range.
- Avoid high traffic areas. Dogs-tails, cats, younger siblings seem to find and spill experiments.
- Shake your cultures.
- If they sink you should resuspend your cultures by shaking them. Sinking is normal.
- The algae tend to settle at the bottom of a container, but it will grow faster if resuspended periodically.
- Shaking also, breaks up the layer of water around each cell, called the “boundary layer”. Shaking breaks up the boundary layer allowing access to nutrients and diffusing wastes (oxygen) from around the cell.
- Every day or every few hours, gently shake the Beaker Bag, stir, or aerate.
- Let the culture breath.
- Open the beaker bag just a bit to let oxygen out and CO2 in and O2 out.
- Day-3-add the rest of the media!
- You were patient and waited for three days- congratulations!
- Make sure the Beaker Bag is fully open, by blowing into it. I know it may look silly, but it works.
- Add the remaining 350mL of media from your 500mL bottle of media you made in step-1.
- Watch the algae grow!
- Quantify biomass growth over time as the culture grows.
- Record the volume, and use the Secchi stick to find the optical density of the algae culture by dipping it into the culture and recording the depth at which you can no longer observe the end. Shake it up right before you make your measurement.
- Youtube video: The Dirty Labcoat: The Secchi Stick
- You can measure every few hours and get results. Seriously- we can get a growth curve by measuring at 8:00 am, 11:00 am, and 2:00 pm. The larger the sample size, the better.
- Leave the bag just barely open to all for gas exchange.Record this data as SDD (Secchi disk depth) over time, then reference the conversion table to convert SDD to cells/mL or cells/volume (CV). Plot CV over time to observe growth graphically.
- Use the attached data sheet to record the secchi disk depth.
- When to stop growing and make beads?
- Every day you can shake up the bag and record the secchi disk depth. You will notice that after 7-14 days the culture will sink overnight. At this point it has exhausted some resources and will sink. The cells are very much still alive.
- The cells will sink when the culture gets to ~20cm of Secchi Disk Depth,
- If the biomass is not getting denser after ~5 days, increase the light available to the culture.
- Once the cultures are sufficiently dense, let it sit overnight without shaking.
- The cells will concentrate at the bottom.
- Gently pour the liquid from the top of the culture (decanting), and you will be left with concentrated cells at the bottom.
- The goal 10-15mL of concentrated algae, let them concentrate again if needed.
- Collect them into the 50-mL self standing centrifuge tube that the original culture came in. We will add the sodium alginate to make the “snot” aka algae-culture and alginate mixture.
Creation of Algae Beads
(Algae Bead Project 2 of 3)
|
Project Summary: This project is for those who wish to create their algae beads, instead of purchasing them whole from Algae Research and Supply. The project can be conducted using cultures provided by ARS or cultures that you yourself have grown.
Time Estimates: ~45 minutes
Learning Outcomes:
Classroom Prep: You will need the algae culture from the previous lesson and the Algae Bead Kit from Algae Research and Supply. Divide class into lab groups if desired. See the Materials section below for the required supplies for this lesson. |
Materials
- Algae culture from the previous lesson,
- Concentrated culture (15mL)
- Algae culture from ARS, (follow procedure to settle/concentrate in from lesson above).
- Make Your Own Algae Beads (A complete kit for growing algae, making algae beads, making pH indicator, and performing experiments.) (ARS sku: ABGK-BB-00050)
- Sodium alginate, dissolved, 15mL
- Dripper
- Calcium chloride, 5g for forming beads
- Spoon or other bead manipulating device
- Chlorine/Calcium-free water (carbon-filtered tap, distilled, or bottled water)
- Safety equipment- glasses, gloves, lab coat, flame thrower (just kidding, really don’t use a flamethrower)
Procedure
- Prepare the algae/alginate solution
- Into the 50mL centrifuge tube, add the algae and sodium-alginate in a 1:1 ratio
- From the previous project, pour your concentrated algae suspension, 15mL into the 50mL centrifuge tube.
- From the 15mL tube of Sodium alginate solution pour the entire tube into the 50mL centrifuge tube with the concentrated algae from the above step. We call this “snot” you will know what we mean after you open and transfer the liquid.
- Mix the algae-alginate solution with the pipette. Try not to get too many bubbles in solution. They will make your beads float. The bubbles will rise out if you leave it for 5-minutes.
- Cap and set this 50mL tube with the solution aside while you make the CaCl2 solution.
- Prepare the hardening solution of calcium chloride, CaCl2
- Rinse out your beaker bag. Completely open the Beaker Bag so it stands up, you may need to blow into it to get it to fully inflate.
- Find the baggie with the white powder labeled CaCl2. Pour the contents of the bag into the Beaker Bag.
- Add 375mL of water to the Beaker bag. This water will dissolve the CaCl2. Ziplock the Beaker Bag to closed and shake it to dissolve. While shaking spin clockwise three times, then counterclockwise six times. Repeat spinning four times. You really do not have spin, but we thought it would entertain your classmates.
- Make beads!
- Note: Do not rush. Do not pour the algae/alginate solution directly into the calcium hardening solution. If you do, the whole mixture will become clumpy and unusable. Although it will look really cool and you will likely be tempted to throw it at a classmate or sibling (don’t get caught).
- Make the beads... drip, drip, drip:
- Open the centrifuge tube with the 50mL of algae-alginate (green-snot) and set it on the bench in front of you.
- Open the Beaker Bag with the calcium chloride solution and set it in front of you as well.
- With the pipette, draw up the algae/alginate solution (green-snot).
- Next slowly drip the solution, drop-by-drop, into the Beaker Bag with the calcium chloride.
- Each drip will form a nearly perfect sphere. The calcium interacts with the sodium alginate and makes a hard bead. You can pull one out to test/look at it after about 20-seconds, but the inside of it will be liquid. We recommend letting them “set” for about 30 minutes.
- Do not submerge the pipette into the hardening solution, it will cause the snot to get hard in and on the pipette- you need only to clean it with water. But it is a hassle to clean stuff all the time, right?
- Try to make the beads be consistent in size, it will make your experimental results be more repeatable .
- Wash the beads
- Pour out the calcium solution from the beaker bag into the sink. Easiest way to do this is to use the zip-loc of the Beaker Bag not completely closed. Inver the bag and let the solution pour out of the bag. Careful not to let the beads out
- Alternatives- Slotted spoon, Tea strainer, small mesh pasta strainer, cheese cloth, window screen.
- Rinse the beads 3x with chlorine/calcium-free water (reverse osmosis or bottled water), then store them in chlorine/calcium-free water for storage until use.
- Cleanup
- Everything can be sent down the drain or into the trash once you’ve completed the procedure.
- DO NOT eat the snot or the algae. It’s not toxic, but it’s also not meant for people to eat. Someone probably needs to read this; you know who you are.
- Wash everything out and you can use it for experiments or other lab work. You will use the 50mL centrifuge tube in next project.
- Storage
- You can store the beads in the Beaker Bag, or self-standing centrifuge tube. Completely submerge them with chlorine/calcium free water.
- Store on a surface out of direct sunlight, but some light is good.
- Refrigeration is not recommended- they are alive!!! Keep the beads at room temperature, in light bright enough to read by, and the cap slightly open to facilitate gas exchange.
- If you see any white precipitate in the bag or on the beads, it is likely calcium from the hardening solution that has combined with carbonate from the water forming calcium carbonate. Simply rinse the beads using the instructions above.
Expected Outcomes
- 150-300 algae beads, depending on the starting volume of concentrated algae.
- Waste products can be disposed of down the sink.
Creation of Algae Bead Vials: pH Indicator Adjustments and Filling Vials
(Algae Bead Project 3 of 3)
|
Project Summary: This project is the last step before doing experiments. These instructions tell you how to fill vials with indicator and algae beads.
Time Estimates: ~45 minutes.
Learning Outcomes:
Classroom Prep: Divide class into lab groups if desired. See the materials list for the required components |
Materials
- Make Your Own Algae Beads (A complete kit for growing algae, making algae beads, making pH indicator, and performing experiments.) (ARS sku: ABGK-BB-00050)
- Dripper
- Indicator, 50ml ready to go
- Microcentrifuge tubes, 1.5mL (x16)
- pH indicator card
- Straw
- Safety equipment- glasses, gloves, lab coat
- Pipette
- Algae beads, from Project-2
- Chlorine/calcium free Water (aka distilled, soft, or reverse osmosis water)
- A few squares of paper towel
- Pair of scissors
Procedure
- Find your pH indicator 50mL, and set it in front of you.
- This tube will be full, if you intend on filling the 16x microcentrifuge tubes full with the beads and indicator. Pour ~25mL of the indicator into the spare cleaned centrifuge tube from the previous project.
- Why- you may need to blow bubbles into it and it can get messy. Also, you will be adding the beads to this tube and do not want to overfill and spill.
- Observe your pH indicator solution. Adjust pH of indicator (you probably won’t need to, but here is how to do it if you want to)
- Caution: Be careful, this has bromothymol blue and crysol red in water and ethanol (alcohol). It will stain clothes and is not good if you ingest it. MDS for carbonate indicator.
- Why are we doing this: Our purpose with the indicator is to observe photosynthesis, which will increase the pH. Therefore, we should start testing the indicator at a low pH (more acidic) by adding carbon dioxide, aka carbonic acid, from exhaled breath.
- Blow bubbles in the indicator using a straw,
- Safety: wear glasses as the indicator can/will splash into your eyes. Better yet, cover the container with a gloved hand and wear glasses. Do not drink it, it does have toxic ingredients, see Caution above.
- Watch the color change, you don’t want to blow past it too far, or the color change will take a long time as photosynthesis will take a while the chew through all the CO2 you blew in.
- We suggest adding CO2 (aka blowing) until the color is just barely out of the brownish and into the yellow. Like this:
- Filling the vials
- Set in front of you
- microcentrifuge vials
- pH indicator filled to ~25mL with pH adjusted solution
- The Beaker Bag with the algae beads
- Fish out of the Beaker Bag around 80 algae beads, set them on a paper towel.
- Civilized people- use a slotted spoon
- The rest of us- use your hand, careful not to spill water everywhere. You may find nervana running your fingers through the beads, try not to make a scene- this is science.
- Add your ~80 algae beads to the centrifuge tube that is filled to ~25mL with the indicator.
- Modify your plastic pipet.
- You are going to use the pipette to transfer beads and indicator solution from the 50mL centrifuge tube into your microcentrifuge tubes.
- The pipette tip is too small for the beads- you will need to make it bigger.
- Use your scissors to cut the tip of the plastic pipette off. Start small and then cut off more. The diameter of the tip should be larger than your largest beads.
- Holding the 50mL centrifuge tube and microcentrifuge tube in one hand. Use the pipette to transfer five (5) algae beads to the smaller tube. We like to use 5, but feel free to add more.
- Cap the tube.
- Repeat for the remaining tubes.
- All done. Store them at room temperature and out of bright light. Pat yourself on the back! Get ready for some fun with experiments!
Page -
Ignite curiosity and foster hands-on learning with our comprehensive algae experiment kits. Designed for educators, students, and science enthusiasts, each kit provides all necessary materials to explore the fascinating world of algae. From culturing diverse algal species to conducting experiments on growth conditions, these kits offer a practical approach to understanding ecological and biological concepts. Ideal for classrooms, science fairs, or personal exploration, our kits make the study of algae accessible and engaging for all ages.