The Biology of Splashpool Copepods (Tigriopus californicus)
BY ALEXIS WIKTOROWICZ-CONROY, PH.D
Life History of Tigriopus californicus
Copepods are so numerous that they have been called “insects of the sea,” representing 70% of the ocean’s biomass and with 12,000 known species described (Raisuddin et al., 2007). Being an intermediary between primary producers (phytoplankton) and larger predators (fishes & filter feeders), copepods are an essential component of the marine food chain (Ruppert et al., 2003). SInce copepods are zooplankton, they are not very strong swimmers and make easy and tasty prey for many fish, inverts, and filter feeders, depending on the life stage of the copepod. Tigriopus californicus is considered meiofauna, inhabiting the substrate and spending most of its time on the benthos looking for detritus. When an adult decides to swim into the water column, its attractive jerky swimming motions create an excited feeding response from predators, making them a great food for picky eaters such as seahorses and mandarin gobies.
Tigriopus californicus, affectionately known as “tig pods,” or “spashpool copepods,” are 2mm-long marine crustaceans belonging to the subclass Copepoda, Order Harpacticoida. T. californicus inhabits splashpools close to the high tidal line along the West coast of North America, from Alaska down to Baja, CA.
A harpacticoid copepod’s development is broken up into several stages (Raisuddin et al., 2007):
- N1-N6: naupliar stages
- C1-C5: copepodid stages
- Adult stage (male or female)

T. californicus developmental stages. Image from Raisuddin et al., 2007.
Life Cycle (Huizinga, 1971)
- A T. californicus egg takes 24 hrs to hatch
- The first stage is a nauplius which molts several times, getting bigger with each molt
- After 5-6 days the nauplius molts into a copepodite which looks like a mini adult
- The copepodite stage lasts from 8-9 days after which the final copepodite turns into an adult copepod
- Males and females are sexually dimorphic; males are slightly smaller (Fraser, 1936)
- After mating, the female lays an egg about 3-4 days later, continually producing egg sacs from store sperm every 2-3 days
- Approximately 18 eggs in an egg sac
- At room temperature the life cycle (from egg to egg) of these copepods is 18-21 days
T. californicus is found in dense numbers in supralittoral pools (splashpools) which are above the spring tidal line, leftover from high tide or storms, replenished by the occasional splashing of waves; these differ from tidepools which are lower in the intertidal (littoral) and whose water is replenished often (Metaxas and Schebling, 1993). Because of the extreme evaporative nature of such pools, the salinity is often quite high; this species of copepod is very hardy and is able to survive in salinities 3 times that of seawater (Meinkoth, 1998). Such splashpools also have a large range of thermal variability and as such these copepods must be able to withstand extreme temperature shifts in addition to salinity shifts (4-40 C; 4-102 ppt; Ranade, 1957; Egloff, 1967; Vittor, 1971). Additionally, there little if not zero migration (gene flow) from one splashpool to the next, nor is there a diapause stage characteristic of other copepods (allowing them to endure unfavorable conditions), so survivability demands on the individual populations are very high- and yet they persist in these extreme circumstances (Burton and Feldman, 1981; Burton, 1997; Edmands and Deimler, 2004; Willett and Ladner, 2009). But despite their hardiness, the region where they hail from dictates their growth rate and fitness; populations from Northern California prefer a lower range of temperatures vs the populations from Southern California, although neither appreciate the extreme ends (Willett, 2010).
In addition to being known for their limited genetic exchange, extreme hardiness, and tastiness to predators, T. californicus has natural sunscreen in its body- a carotenoid called astaxanthin, serving as protection from the sun’s photo-oxidative effects (Hylander et al., 2015; Weaver et al., 2018; Raisuddin et al., 2007). Supralittoral pools receive high amounts of UV, and studies have shown T. californicus may accumulate more astaxanthin in its carapace when exposed to higher amounts of UV-B (Chalker-Scott, 1995), while in another copepod, Tigriopus brevicornis, it has been verified (Davenport et al., 2004). And just as a mother (ok, dads too) lathers up her children with sunscreen, a female copepod passes on her astaxanthin to her young through her eggs (Chalker-Scott, 1995).
References:
Burton, R.S. 1997. Genetic evidence for long term persistence of marine invertebrate populations in an ephemeral environment. Evolution. 51: 993–998.
Burton, R.S., and M.W. Feldman. 1981.Population genetics of Tigriopus californicus. II. Differentiation among neighboring populations. Evolution. 35:1192–1205.
Chalker-Scott, L. 1995. Survival and sex ratios of the marine copepod, Tigriopus californicus, following ultraviolet-B (290-320 nm) radiation exposure. Marine Biology. 123: 799-804.
Davenport, J., Healy, A., Casey, N., J.J.A Heffron. 2004. Diet-dependent UVAR and UVBR resistance in the high shore harpacticoid copepod Tigriopus brevicornis. Marine Ecology Progress Series. 276: 299-303.
Edmands, S., and J.K. Deimler. 2004. Local adaptation, intrinsic coadaptation and the effects of environmental stress on interpopulation hybrids in the copepod Tigriopus californicus. Journal of Experimental Marine Biology and Ecology. 303: 183–196.
Egloff, D. A. 1966. Ecological aspects of sex ratio and reproduction in experimental and field populations of the marine copepod Tigriopus californicus. Ph.D. Dissertation, Stanford Univ., Palo Alto, CA .
Frasier, J.H. 1936. The occurrence, life history, and life history of Tigriopus fulvus (Fischer). Journal of the Marine Biological Association of the U.K. 20: 523-536.
Huizinga, H.W. 1971. Cultivation, life history and salinity tolerance of the tidepool copepod, Tigriopus californicus Baker 1912, in artificial sea water. Transactions Illinois Academy of Science. 64: 230-236.
Hylander, S., Kiørboe, T., Snoeijs, P., Sommaruga, R., T.G. Nielsen. 2015. Concentrations of sunscreens and antioxidant pigments in Arctic Calanus spp. In relation to ice cover, ultraviolet radiation, and the phytoplankton spring bloom. Limnology & Oceanography. 60: 2197-2206.
Meinkoth, N.A. 1998. National Audubon Society Field Guide to North American Seashore Creatures. Chanticleer Press, Inc., New York, USA.
Metaxas A., and R.E. Scheibling. 1993. Community structure and organization of tidepools. Marine Ecology Progress Series. 98: 187-198.
Raisuddin, S., Kwok, K.W.H., Leung, K.M.Y., Schlenk, D. and J.S. Lee. 2007. The copepod Tigriopus: A promising marine model organism for ecotoxicology and environmental genomics. Aquatic Toxicology. 83: 161-173.
Ranade, M.R. 1957. Observations on the resistance of Tigriopus fulvus (Fischer) to changes in temperature and salinity. Journal of the Marine Biological Association of the U.K. 36: 115-119.
Ruppert, E.E., Fox, R.S., Barnes, R.D., 2003. Invertebrate Zoology. A Functional
Evolutionary Approach, 7th ed. Brooks/Cole-Thomson Learning, Belmont, CA, USA.
Weaver, R.J., Cobine, P.A., and G.E.Hill. 2018. On the bioconversion of dietary carotenoids to astaxanthin in the marine copepod, Tigriopus californicus. Journal of Plankton Research. https://doi.org/10.1093/plankt/fbx072
Willett, C.S. 2010. Potential fitness trade-offs for thermal tolerance in the intertidal copepod Tigriopus californicus. Evolution. 64: 2521-2534.
Willett, C.S., and J.T. Ladner. 2009. Investigations of fine‐scale phylogeography in Tigriopus californicus reveal historical patterns of population divergence. BMC Evolutionary Biology. 9: 139.
Vittor, B.A. 1971. Effects of the environment on fitness‐related life history characters in Tigriopus californicus. PhD dissertation, Univ. of Oregon, Eugene.
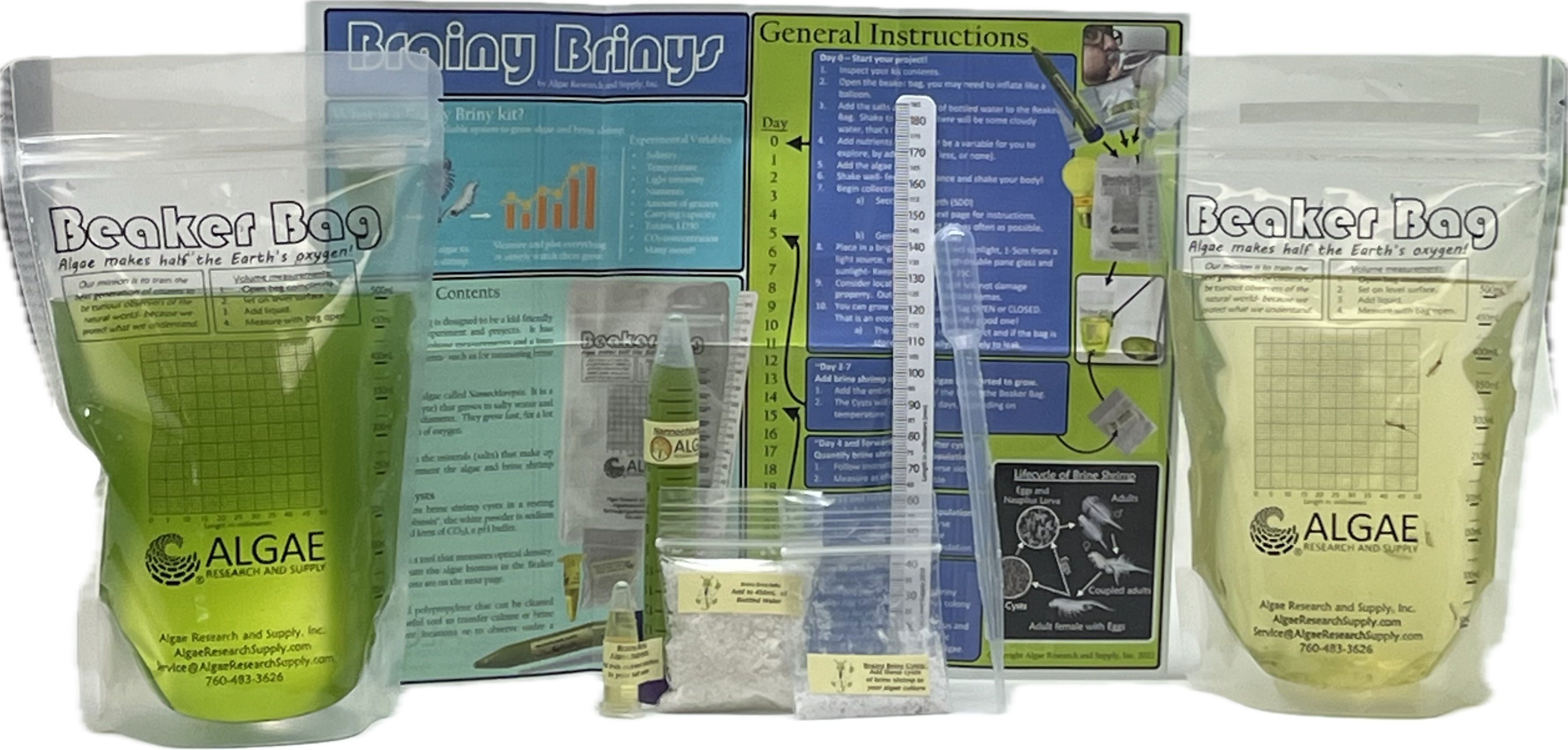
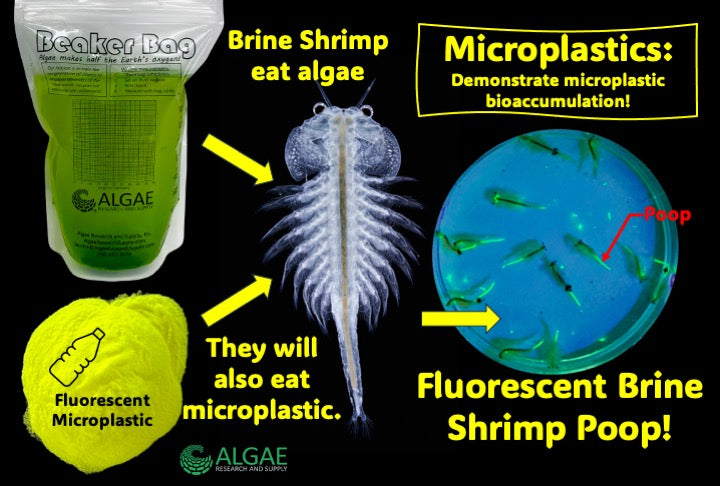
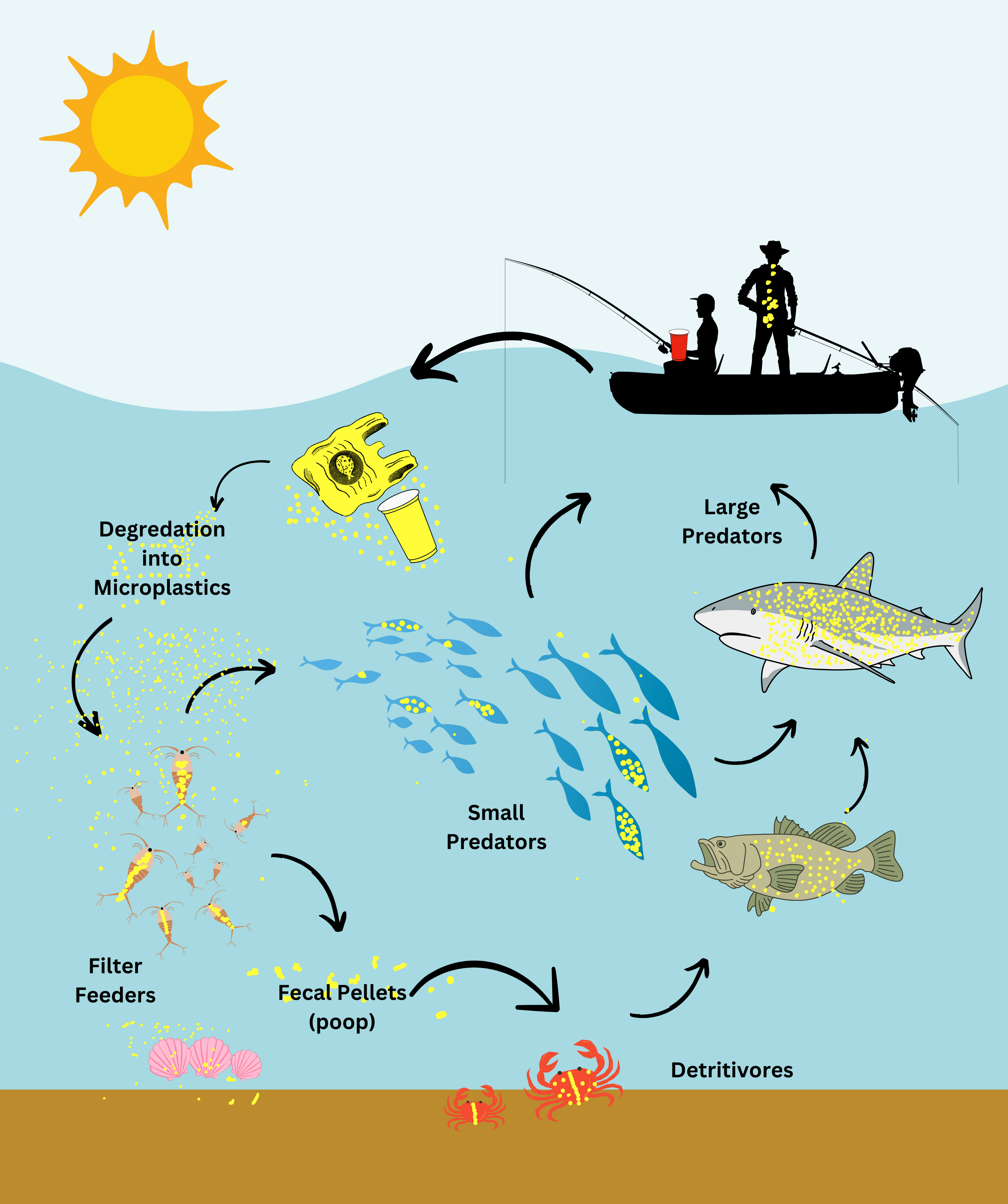
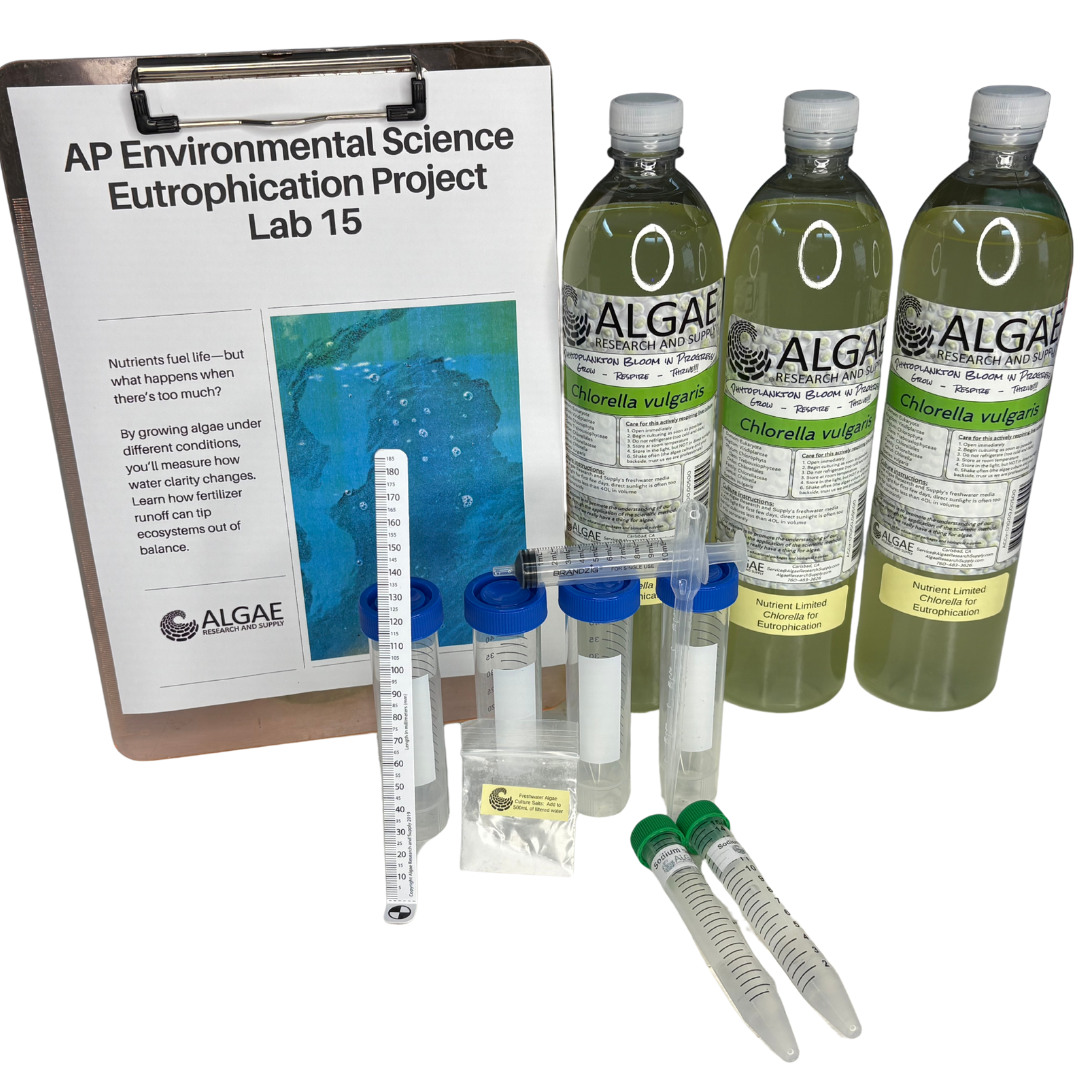
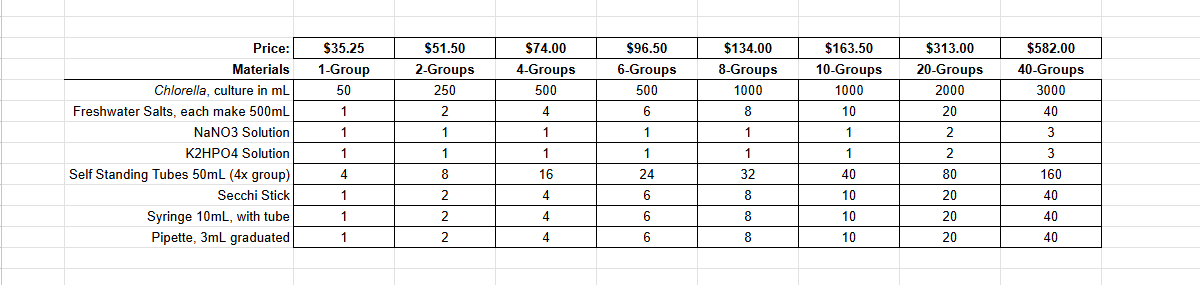
Ignite curiosity and foster hands-on learning with our comprehensive algae experiment kits. Designed for educators, students, and science enthusiasts, each kit provides all necessary materials to explore the fascinating world of algae. From culturing diverse algal species to conducting experiments on growth conditions, these kits offer a practical approach to understanding ecological and biological concepts. Ideal for classrooms, science fairs, or personal exploration, our kits make the study of algae accessible and engaging for all ages.